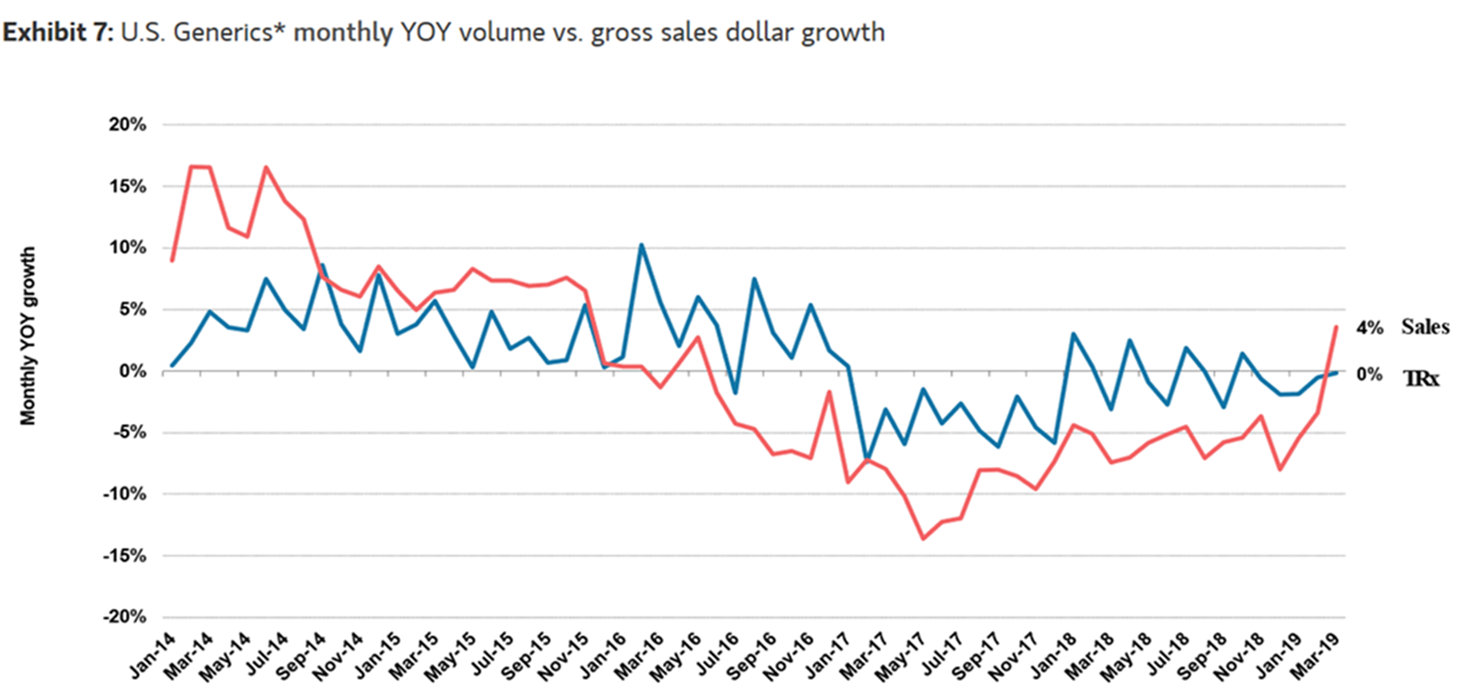
WASHINGTON, DC (May 12, 2019) — The Association for Accessible Medicines (AAM) and its member companies are committed to supporting policies that promote competition and help speed the availability of generic and biosimilar medicines to patients. Today’s generic drug industry is characterized by intense competition. As a result, pricing data from the last three years indicate that generic prices have declined overall and saved patients and taxpayers literally billions of dollars compared to brand-name drug prices. (See chart below – source IQVIA, Morgan Stanley)
AAM is fully committed to compliance with all laws and to maintaining high ethical standards in the way we do business. Illegal behavior, such as price-fixing or other violations of antitrust law, is inconsistent with AAM’s rules and procedures. We seek to maintain the highest ethical standards as we work to bring medicine to patients in the U.S.
AAM has an established Code of Business Ethics, which reflects international principles of business ethics, including the Asia Pacific Economic Cooperation (APEC) Mexico City Principles on Voluntary Codes of Business Ethics in the Biopharmaceutical Sector. We have established procedures and policies to help ensure compliance with the antitrust rules at our board meetings and all other AAM meetings. We will continue to assess our antitrust policies because we are committed to the idea that robust competition is the key to providing affordable and accessible medicines to patients while also constraining health care costs.
Generic drug and biosimilar developers support laws and regulations that promote competition. The best way to lower and control drug costs is through the promotion of policies that incentivize competition and bring more generics to the market. As the pharmaceutical industry data tracker, IQVIA, has just reported, generic medicines account for 90% of the prescriptions dispensed in this country, yet represent only 22% of all drug spending, and 95% of generic prescriptions are filled for $20 or less. Over the past decade, the U.S. health care system has saved $2 trillion due to the availability of low cost generics. In 2018, generics generated a total of $293 billion in savings. Savings for Medicare and Medicaid amounted to $90.3 billion and $46.8 billion, respectively, which translates to an average $2,254 for every Medicare enrollee and $817 for every Medicaid enrollee.
Generic drug savings provide the health care system with the ability to invest in new medications. AAM will continue to support policies that promote competition and help speed the availability of generic medicines to patients.
MEDIA CONTACT:
Rachel Schwartz
202.249.7147
About AAM
AAM is driven by the belief that access to safe, quality, effective medicine has a tremendous impact on a person’s life and the world around them. Generic and biosimilar medicines improve people’s lives, improving society and the economy in turn. AAM represents the manufacturers and distributors of finished generic pharmaceuticals and biosimilars, manufacturers and distributors of bulk pharmaceutical chemicals, and suppliers of other goods and services to the generic industry. Generic pharmaceuticals are 90 percent of prescriptions dispensed in the U.S. but only 22 percent of total drug spending.
About the Biosimilars Council
The Biosimilars Council, a division of the Association for Accessible Medicines (AAM), works to ensure a positive environment for patient access to biosimilar medicines. The Biosimilars Council is a leading source for information about the safety and efficacy of more affordable alternatives to costly brand biologic medicines. Areas of focus include public and health expert education, strategic partnerships, government affairs, legal affairs and regulatory policy. More information is available at www.biosimilarscouncil.org.